Why MTD
Experts in Problem Solving
Problem solving is what drives the healthtech and medical device industry.
For OEMs to successfully produce medical solutions, they often need to overcome manufacturing challenges. Whether manufacturing a micro component in-house or working with another molding supplier, sometimes OEMs hit roadblocks.
Sometimes the molder simply doesn’t have the specialized medical micro molding equipment needed to produce that particular component. Sometimes they don’t have experience or expertise in working with a certain material or challenging design. Other times, a supplier may excel at low volume or on-demand production, but cannot efficiently ramp up production.
Regardless of what project phase you’re in, it’s never too late to talk to MTD. We can help get your product development back on track and to market faster and more cost-effectively.
Because all our services are under one roof, our engineers can efficiently resolve problems and challenges. With MTD’s micro medical molding process, quality standards and sophisticated equipment working together, we help customers push the envelope— allowing predictable outcomes even on breakthrough products that were never produced before.
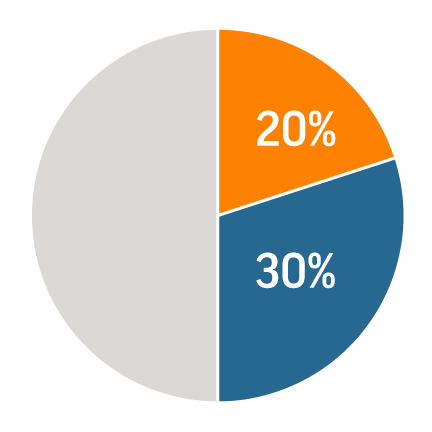
20% of new projects come to MTD as "rescues," or failed attempts by others.
Another 30% are projects no one else would even attempt.
Top Micro Molding Issues
About 80% of the problems that MTD helps customers resolve fall into five areas.
No one else can do or will quote
- Told design is “impossible”
- Asked to make numerous or significant design compromises
Part quality issues
- Part-to-part or lot-to-lot inconsistency – metrology, mechanical failures, dimensional
- Low part quality or dimensional stability
- Timely, costly, and/or inaccurate assembly steps
Bioabsorbable molding expertise needed
- Bioabsorbable IV inconsistency
- Bioabsorbable prototyping
In-house (or supplier) manufacturing issues
- Inadequate material characterization and selection
- Polymer analysis capabilities needed
- Part/process validation required
- Unique assembly required
- Custom packaging needed
Route for production scalability needed
- Material inefficiency – issues with material waste
- Supplier consolidation needed
- Higher production costs
- Weak supply chain security
Case Studies
Keys to Bioabsorbable Process Development:
Consistency & Control
A medical device company developed a bioabsorbable fixation design and concept that was celebrated by surgeons. They worked with a reputable molder, but after several years, the molder had limited success and was unable to produce the part represented in the drawing. By guiding the customer through material characterization and developing the unique tooling construction concept to reduce secondary operations, MTD's micromolded parts achieved minimal and consistent IV loss and were much more consistent shot to shot. This confidence in the repeatability of device function allowed reliable testing and successful proof of concept to take place.

